Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Yamauchi, Akihiro*; Amaya, Masaki
Proceedings of Annual Topical Meeting on Reactor Fuel Performance (TopFuel 2018) (Internet), 7 Pages, 2018/10
Differential scanning calorimetry (DSC) measurements on pre-hydrided cold worked, stress relieved and recrystallized Zry-4 cladding were performed in a temperature range between 50 and 600 C in order to elucidate the effect of final heat treatment at fabrication of Zircaloy-4 (Zry-4) cladding on the terminal solid solubility during the dissolution of zirconium hydrides at heating up (TSSD). Obtained DSC curves and Metallography indicate that the initial state of hydrides affects the dissolution behavior of hydride. The Arrhenius plots of the TSSD temperatures and hydrogen contents obtained from this study revealed that cold worked samples exhibited the largest TSSD and followed by stress relieved and recrystallized samples. The results of this study indicated that the difference in microstructure due to final heat treatment at fabrication of Zry-4 cladding affects the dissolution behavior of hydrides.
C in order to elucidate the effect of final heat treatment at fabrication of Zircaloy-4 (Zry-4) cladding on the terminal solid solubility during the dissolution of zirconium hydrides at heating up (TSSD). Obtained DSC curves and Metallography indicate that the initial state of hydrides affects the dissolution behavior of hydride. The Arrhenius plots of the TSSD temperatures and hydrogen contents obtained from this study revealed that cold worked samples exhibited the largest TSSD and followed by stress relieved and recrystallized samples. The results of this study indicated that the difference in microstructure due to final heat treatment at fabrication of Zry-4 cladding affects the dissolution behavior of hydrides.
Ioka, Ikuo; Suga, Masataka*; Nagase, Fumihisa; Futakawa, Masatoshi; Kiuchi, Kiyoshi
JAERI-Tech 2001-013, 111 Pages, 2001/03
no abstracts in English
Watanabe, Kazuo
Hyojun Busshitsu; Bunseki, Keisoku No Shinraisei Kakuho No Tameni, p.175 - 178, 1998/00
no abstracts in English
Watanabe, Kazuo
Bunseki, 0(3), p.196 - 197, 1996/00
no abstracts in English
;
Bunseki Kagaku, 43, p.489 - 494, 1994/00
no abstracts in English
; ; ; ; ; Shibahara, Itaru
PNC TN9410 92-321, 30 Pages, 1992/10
Integrity evaluations have been performed for the 2nd Fugen pressure tube test (8 years irradiation, 5.6  10
10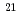 n/cm
n/cm (E
(E 1Mev)). Test items mainly consist of tensile test, bending test, corrosion test and hydrogen analysis. It has become clear using these data that the pressure tube material has maintained its integrity during the irradiation by the integrity assessment on both tensile and fracture toughness properties. Besides, both thickness loss by corrosion and absorbed hydrogen content were lower than those of design values.
1Mev)). Test items mainly consist of tensile test, bending test, corrosion test and hydrogen analysis. It has become clear using these data that the pressure tube material has maintained its integrity during the irradiation by the integrity assessment on both tensile and fracture toughness properties. Besides, both thickness loss by corrosion and absorbed hydrogen content were lower than those of design values.
JAERI-M 91-044, 85 Pages, 1991/03
no abstracts in English
Kato, Kaneharu
Bunseki Kagaku, 39(8), p.439 - 444, 1990/08
no abstracts in English
*; Kawamura, Hiroshi; *;
Nihon Seramikkusu Kyokai Gakujutsu Rombunshi, 98(1139), p.721 - 725, 1990/07
no abstracts in English
JAERI-M 85-028, 43 Pages, 1985/03
no abstracts in English
JAERI-M 83-241, 64 Pages, 1984/02
no abstracts in English
Miura, Makoto; ; ; Omori, Takuro; ; Tanaka, Yasumasa; Shiina, Sadamu; Ogasawara, Koji
PNC TN841 79-12, 103 Pages, 1979/03
no abstracts in English
; *
Bunseki Kagaku, 19(3), p.403 - 406, 1970/00
no abstracts in English
Nakahara, Kaito*; Kawada, Rio*; Irisawa, Eriko; Ueda, Mitsutoshi*; Kawamura, Kenichi*
no journal, ,
In order to develop oxygen sensor using oxide scales formed on the surface of zirconium alloy, we focused on zirconium and copper alloy system. It was expected that the oxide scale of Zr oxide on the alloy is thin and elaborate compared with that on the pure Zr because the activity of Zr in the alloy is reduced and the oxidation rate of alloy become slow. Therefore the oxidation behavior and mechanism of two phase alloy, Zr-35Cu which is composed of Zr Cu and Zr
Cu and Zr Cu
Cu , was studied. The results showed the oxidation rate of the alloy was same with that of the pure Zr.
, was studied. The results showed the oxidation rate of the alloy was same with that of the pure Zr.
Ohigashi, Jun*; Kawamura, Kenichi*; Ueda, Mitsutoshi*; Irisawa, Eriko; Komatsu, Atsushi
no journal, ,
In order to apply zirconium alloys and their oxide films to oxygen sensors, high temperature oxidation experiments were conducted on Zr-63 at.%Al, Zr-39 at.%Al, and Zr-31 at.%Y alloys. The oxidation behavior of these alloys was observed, and the oxide ion conductivity was examined by measuring the electromotive force of the oxide films of the alloys whose oxidation inhibition was confirmed. Zr-39 at.% Al and Zr-31 at.% Y alloys. On the other hand, a thin, uniform, dense zirconium oxide film was formed for the Zr-63 at.%Al alloy. The electromotive force (EMF) of this oxide film was measured in a gas of known oxygen concentration, but it was smaller than the expected EMF, and the oxide ion conductivity was not sufficient.